Image source: Shutterstock
Highlights
- NEU reported a 594% YoY increase in royalty revenue in 1HFY24, reaching AUD 24.3 million.
- Acadia Pharmaceuticals submitted a Marketing Authorization Application to the European Medicines Agency for trofinetide to treat Rett syndrome.
- NEU received its first sales milestone payment of USD 50 million after DAYBUE™ exceeded USD 250 million in net sales.
Neuren Pharmaceuticals Limited (ASX:NEU) is an Australian company that focuses on developing innovative drug therapies for neurological disorders. The company’s product, DAYBUE™ (trofinetide), has received approval from the US FDA.
Through the commercial success of DAYBUE™, the company expects to benefit from ongoing royalties and sales milestone payments. The company expects to see revenue growth with the expansion of markets in North America, Europe, and Canada.
NEU partnered with Acadia Pharmaceuticals (NASDAQ:ACAD), offering a foundation for continuous revenue growth.
Trofinetide Marketing Authorization Application for European Approval
On 15 January 2025, NEU revealed that its partner, Acadia Pharmaceuticals has submitted a Marketing Authorization Application (MAA) to the European Medicines Agency (EMA) for the use of trofinetide as a treatment for Rett syndrome in patients aged two years and older.
If approved, trofinetide would become the first authorized therapy for Rett syndrome in the European Union. Acadia anticipates receiving regulatory approval in the first quarter of 2026. Managed Access Programs are expected to launch across Europe by the second quarter of 2025.
3QFY24 Financial Performance Update
In the third quarter of the financial year 2024 (3QFY24), royalty income reached AUD 13.2 million, contributing to a total of AUD 37.5 million for the first nine months of the fiscal year.
DAYBUETM achieved net sales exceeding USD 250 million in 3QFY24, triggering NEU’s first sales milestone payment of USD 50 million. Acadia Pharmaceuticals reported third-quarter sales of USD 91.2 million, reflecting an 8% growth from the previous quarter and a 36% YoY increase.
1HFY24 Financial Performance Update
In 1HFY24, NEU recorded a significant increase in royalty revenue, rising 594% YoY to AUD 24.3 million compared to AUD 3.5 million in 1HFY23. However, this growth was offset by the absence of milestone payments, which had significantly contributed to earnings in the prior period.
As a result, net profit sharply declined by 83% YoY, from AUD 47.8 million in 1HFY23 to AUD 8.0 million in 1HFY24. At the same time, cash and cash equivalents decreased by 93% YoY, from AUD 38.4 million to AUD 2.6 million during the same period.
During the reporting period, NEU remained focused on advancing its clinical programs, including the Phase 2 trials for NNZ-2591. The company is also preparing for Phase 3 development of this candidate, targeting multiple indications.
FY24 Income Guidance
NEU forecasts a total income range of AUD 216 million to AUD 218 million for FY24, encompassing royalties, sales milestone payments, share of PRV net proceeds, and interest income. Royalties for Q4 are expected to fall between AUD 16 million and AUD 18 million.
Expansion of DAYBUET™ in North America
With the recent approval of DAYBUET™ for Rett syndrome by Health Canada, NEU will include Canadian net sales in its royalty calculations alongside US sales. This expansion is anticipated to boost future royalties and milestone payments, with the potential to reach up to USD 350 million based on sales performance.
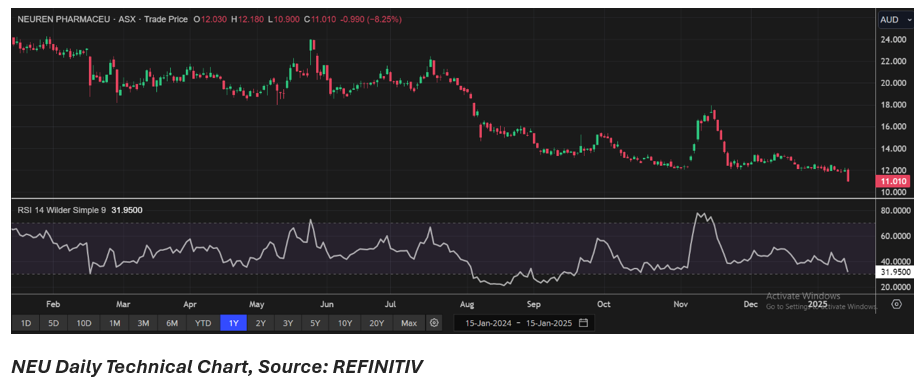
Share performance of NEU
NEU shares closed 8.25% lower at AUD 11.01 per share on 15 January 2025 with AUD 1.56 billion market cap. Over the past year, the share price has declined by almost 53.58% and in three months, it has dropped by 13.31%.
52-week high of NEU is AUD 24.25, recorded on 30 January 2024 and 52-week low is AUD 11.75, recorded on 13 January 2025.
Note 1: Past performance is neither an Indicator nor a guarantee of future performance.
Note 2: The reference date for all price data, and currency, is 15 January 2025. The reference data in this report has been partly sourced from REFINITIV.







 Please wait processing your request...
Please wait processing your request...