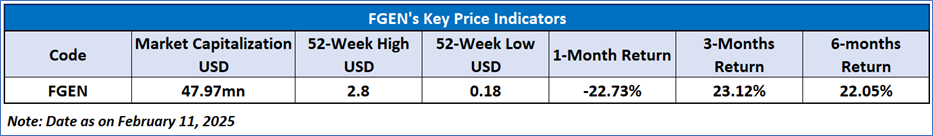
FibroGen Inc
FibroGen, Inc. (NASDAQ: FGEN) is a biopharmaceutical company. The Company is engaged in developing and commercializing a diversified pipeline of novel therapeutics that work at the frontiers of cancer biology and anemia. Its clinical, commercial, and research programs include Pamrevlumab, which is a first-in-class antibody being developed by FibroGen to inhibit the activity of connective tissue growth factor (CTGF).
Recent Business and Financial Updates
- Clinical Development Updates: FibroGen, Inc. has announced that topline results from the Phase 2 portion of the investigator-sponsored study of FG-3246, a first-in-class antibody-drug conjugate (ADC) targeting CD46, in combination with enzalutamide for patients with metastatic castration-resistant prostate cancer (mCRPC), are anticipated in the first half of 2025. Furthermore, the company plans to initiate a Phase 2 monotherapy dose optimization study of FG-3246 in mCRPC in the first quarter of 2025.
- Financial Performance: The company reported a 15% year-over-year increase in third-quarter net revenue, primarily attributed to the strong performance of roxadustat in China, which recorded a 34% growth in sales volume. FibroGen reaffirmed its full-year net product revenue guidance of USD135 million to USD150 million, with total projected roxadustat net sales in China expected to range between USD330 million and USD350 million.
- Operational Efficiency and Cost Reduction: FibroGen has made substantial progress in its U.S. cost reduction initiative, which is anticipated to be largely completed by the end of 2024. This strategic transformation is aimed at ensuring long-term financial stability and sustainable growth.
- Financial Position: As of September 30, 2024, FibroGen reported cash, cash equivalents, and accounts receivable amounting to USD160.0 million. The company expects that, with additional repatriation of cash from its China operations, its cash reserves will be adequate to support operational plans through 2026.
- Management Commentary: Chief Executive Officer Thane Wettig emphasized the company’s transition into a more streamlined and focused organization, resulting in significant cost savings. He underscored the continued strong performance of roxadustat, which generated USD96.6 million in net sales in China during the third quarter. Wettig further reaffirmed FibroGen’s commitment to advancing FG-3246, with key milestones including topline results from the Phase 2 combination study at the University of California, San Francisco (UCSF) and the planned initiation of a Phase 2 monotherapy trial in early 2025.
- Recent Clinical and Regulatory Developments: FibroGen disclosed topline results from the pamrevlumab arm of the PanCAN Precision Promise Phase 2/3 adaptive platform trial for metastatic pancreatic ductal adenocarcinoma (mPDAC), which did not meet the primary endpoint. Similarly, the company reported results from the LAPIS Phase 3 study of pamrevlumab in patients with locally advanced, unresectable pancreatic cancer (LAPC), which also did not meet the primary endpoint.
- Upcoming Milestones: FibroGen expects a regulatory decision on roxadustat for chemotherapy-induced anemia (CIA) in China in early 2025. If approved, the company will receive a USD10 million milestone payment from AstraZeneca. Topline results from the Phase 2 portion of the investigator-sponsored Phase 1b/2 study of FG-3246 in combination with enzalutamide in patients with mCRPC are anticipated in the first half of 2025. Additionally, the Phase 2 monotherapy dose optimization study, which includes a sub-study of FG-3180 to assess CD46 expression and response to FG-3246, is expected to commence in the first quarter of 2025.
- Market Performance in China: FibroGen’s third-quarter net product revenue under U.S. GAAP from roxadustat sales in China amounted to USD46.2 million, reflecting a 57% increase compared to the third quarter of 2023. Total roxadustat net sales in China, including those from the joint distribution entity with AstraZeneca, reached USD96.6 million, marking a 25% year-over-year growth, largely driven by a 34% increase in sales volume. Roxadustat remains the leading brand in the anemia of chronic kidney disease (CKD) market in China.
- Financial Highlights: Total revenue for the third quarter of 2024 was USD46.3 million, representing a 15% year-over-year increase from USD40.1 million in the third quarter of 2023. Net loss for the third quarter of 2024 was USD17.1 million, or USD0.17 per basic and diluted share, compared to a net loss of USD63.6 million, or USD0.65 per basic and diluted share, in the same period of the prior year. The company expects its current cash position and additional cash repatriation from its China operations to support operational plans through 2026.
Technical Observation (on the daily chart):
FGEN's stock has faced resistance at crucial levels and has experienced a decline, suggesting a potential consolidation phase with a bearish outlook. The 14-day Relative Strength Index (RSI) is presently below the midpoint, further reinforcing the negative sentiment at these levels. Additionally, the stock is nearing the point of dropping below its 50-period simple moving average, which would further bolster the negative bias.
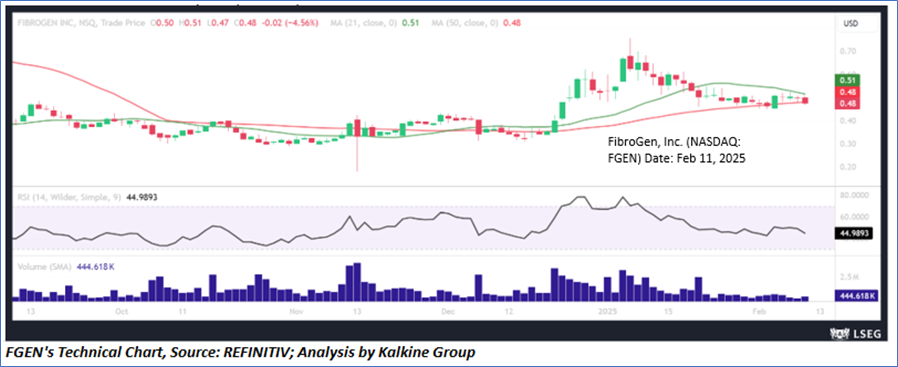
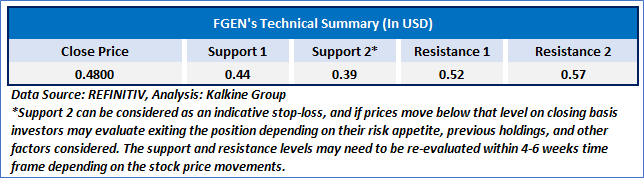
As per the above-mentioned price action, recent key business and financial updates, momentum in the stock over the last month, and technical indicators analysis, a ‘WATCH`’ rating has been given for FibroGen, Inc. (NASDAQ: FGEN) at the close price of USD 0.48 as of February 11, 2025.
Individuals can evaluate the stock based on the support and resistance levels provided in the report in case of keen interest taking into consideration the risk-reward scenario.
Markets are trading in a highly volatile zone currently due to certain macro-economic issues and prevailing geopolitical tensions. Therefore, it is prudent to follow a cautious approach while investing.
Related Risk: This report may be looked at from a high-risk perspective and a recommendation is provided for a short duration. This report is solely based on technical parameters, and the fundamental performance of the stocks has not been considered in the decision-making process. Other factors which could impact the stock prices include market risks, regulatory risks, interest rates risks, currency risks, social and political instability risks etc.
Note 1: Past performance is not a reliable indicator of future performance.
Note 2: The reference date for all price data, currency, technical indicators, support, and resistance level is February 11, 2025. The reference data in this report has been partly sourced from REFINITIV.
Note 3: Investment decisions should be made depending on an individual's appetite for upside potential, risks, holding duration, and any previous holdings. An 'Exit' from the stock can be considered if the Target Price mentioned as per the Valuation and or the technical levels provided has been achieved and is subject to the factors discussed above.
Note 4: Target Price refers to a price level that the stock is expected to reach as per the relative valuation method and or technical analysis taking into consideration both short-term and long-term scenarios.
Note 5: ‘Kalkine reports are prepared based on the stock prices captured either from the New York Stock Exchange (NYSE), NASDAQ Capital Markets (NASDAQ), and or REFINITIV. Typically, all sources (NYSE, NASDAQ, or REFINITIV) may reflect stock prices with a delay which could be a lag of 15-20 minutes. There can be no assurance that future results or events will be consistent with the information provided in the report. The information is subject to change without any prior notice.
AU




 Please wait processing your request...
Please wait processing your request...