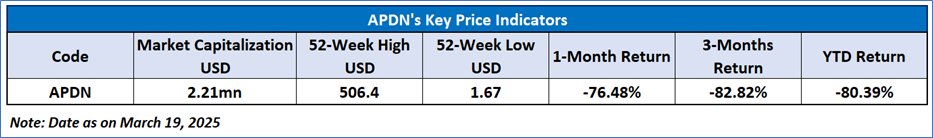
Applied DNA Sciences Inc
Applied DNA Sciences, Inc. (NASDAQ: APDN) is a biotechnology company. The Company is engaged in developing and commercializing technologies to produce and detect deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). The Company operates through three segments: Therapeutic DNA Production Services, MDx Testing Services, and DNA Tagging and Security Products and Services.
Recent Business and Financial Updates
- Overview of Applied DNA Sciences’ First Quarter Fiscal 2025 Results and Strategic Updates: Applied DNA Sciences, Inc. (NASDAQ: APDN), a leader in PCR-based DNA technologies, announced its financial results for the first quarter of fiscal 2025, ending December 31, 2024, on February 13, 2025. The company reported a 34% increase in total revenues, reaching USD 1.2 million compared to USD 891 thousand in the same period of fiscal 2024, alongside a strategic restructuring that included exiting its DNA Tagging and Security Products and Services business segment. Key operational advancements included the completion and certification of its GMP Site 1 facility in Stony Brook, New York, and a workforce reduction of approximately 20%, aimed at optimizing financial stability and focusing on synthetic DNA manufacturing.
- Strategic Restructuring and Corporate Developments: In December 2024, Applied DNA initiated a strategic restructuring, culminating in the announced exit from its DNA Tagging business segment in early 2025, while continuing to service select existing customer contracts. This restructuring involved a workforce reduction of 20%, primarily affecting the DNA Tagging segment, expected to reduce annual payroll costs by 13%, though offset by one-time separation costs of USD 300,000 to be recorded in the quarter ending March 31, 2025. Dr. James A. Hayward, Chairman and CEO, emphasized that these measures are critical to lowering the company’s cash burn rate and stabilizing its financial position to support near-term operational goals.
- Advancements in LineaRx and GMP Facility Operations: The LineaRx subsidiary, focused on therapeutic DNA production, achieved a significant milestone with the completion of its initial GMP facility, Site 1, in Stony Brook, New York, on January 31, 2025. Certified for commercial operation with ISO 7- and ISO 5-compliant workspaces, Site 1 has an initial manufacturing capacity of approximately ten grams of Linea DNA™ IVT templates per annum, potentially generating annual revenues of USD 10 million to USD 30 million, depending on product mix. The facility enables the company to serve clients across all product stages—RUO, GLP, and GMP—supporting research, pre-clinical, and clinical phases, with a first GMP order for IVT templates anticipated in the quarter ending June 30, 2025, pending successful process development with a U.S.-based therapeutics developer.
- Clinical Validation and Expansion of MDx Testing Services: Applied DNA’s Linea DNA platform achieved its first-in-human clinical validation through its use in a CAR-T therapy under a Phase I clinical trial approved by the Czech Republic State Institute for Drug Control, sponsored by the Institute of Hematology and Blood Transfusion. This milestone highlights Linea DNA’s capability to accelerate manufacturing timelines compared to traditional plasmid DNA-based therapies, positioning the company as a leader in enzymatic DNA production. Additionally, the Applied DNA Clinical Labs subsidiary expanded its TR8™ PGx testing service to all U.S. states recognizing New York’s CLEP/CLIA certification, with samples from outside New York to be processed at its Stony Brook diagnostic laboratory, enhancing its market reach.
- Financial Performance and Liquidity Position: For the first quarter of fiscal 2025, Applied DNA reported an operating loss of USD 3.0 million, an improvement from USD 3.8 million in the prior year, while the net loss increased to USD 2.7 million from USD 1.1 million, reflecting restructuring costs and strategic shifts. Adjusted EBITDA improved to negative USD 2.9 million from negative USD 3.2 million, indicating better operational efficiency. As of December 31, 2024, the company’s cash and cash equivalents stood at USD 9.3 million, bolstered by USD 5.7 million in net proceeds from a registered direct offering closed on October 31, 2024, providing a solid foundation to support its focus on commercializing GMP Site 1 and advancing its synthetic DNA production strategy.
- Outlook and Competitive Positioning: Hayward underscored the company’s focus on commercializing the DNA production capacity of GMP Site 1, aiming to secure initial orders for clinical-grade materials to validate its low-CAPEX enzymatic DNA production approach, which offers a cell-free, scalable alternative to traditional plasmid DNA processes. This capability is seen as a unique competitive advantage in the genetic medicines market, where rapid development is critical. With the strategic exit from the DNA Tagging segment and enhanced operational focus, Applied DNA is poised to leverage its Linea DNA platform to drive growth and establish itself as a key player in the synthetic DNA manufacturing industry.
Technical Observation (on the daily chart):
The 14-day Relative Strength Index (RSI) is currently at 19.85, currently inside the oversold zone, with the expectations of consolidation or a reversal if USD 1.70-USD 2.00 levels hold. In addition, the current price is below both the 50-day and 200-day Simple Moving Averages (SMAs), which may work as medium to long term resistance levels.
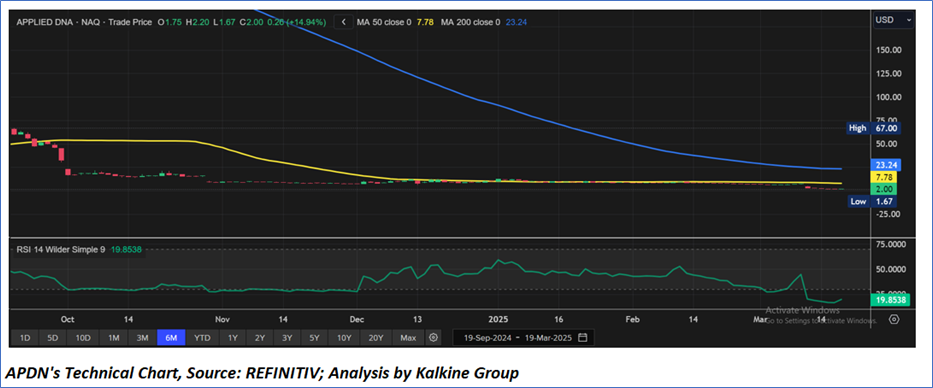
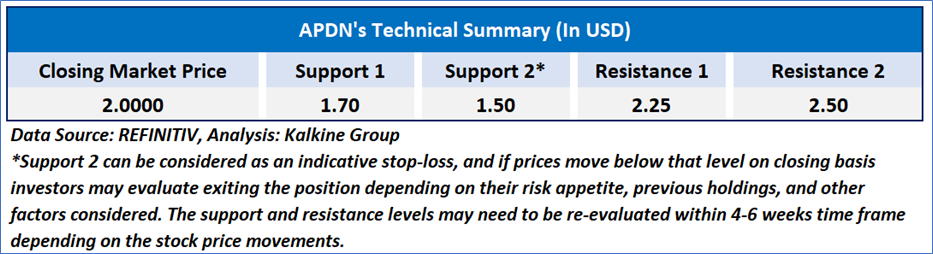
As per the above-mentioned price action, recent key business and financial updates, momentum in the stock over the last month, and technical indicators analysis, a ‘WATCH’ rating has been given Applied DNA Sciences, Inc. (NASDAQ: APDN) at the closing market price of USD 2.00, as of March 19, 2025.
Individuals can evaluate the stock based on the support and resistance levels provided in the report in case of keen interest taking into consideration the risk-reward scenario.
Markets are trading in a highly volatile zone currently due to certain macro-economic issues and prevailing geopolitical tensions. Therefore, it is prudent to follow a cautious approach while investing.
Related Risk: This report may be looked at from a high-risk perspective and a recommendation is provided for a short duration. This report is solely based on technical parameters, and the fundamental performance of the stocks has not been considered in the decision-making process. Other factors which could impact the stock prices include market risks, regulatory risks, interest rates risks, currency risks, social and political instability risks etc.
Note 1: Past performance is not a reliable indicator of future performance.
Note 2: The reference date for all price data, currency, technical indicators, support, and resistance level is March 19, 2025. The reference data in this report has been partly sourced from REFINITIV.
Note 3: Investment decisions should be made depending on an individual's appetite for upside potential, risks, holding duration, and any previous holdings. An 'Exit' from the stock can be considered if the Target Price mentioned as per the Valuation and or the technical levels provided has been achieved and is subject to the factors discussed above.
Note 4: Target Price refers to a price level that the stock is expected to reach as per the relative valuation method and or technical analysis taking into consideration both short-term and long-term scenarios.
Note 5: ‘Kalkine reports are prepared based on the stock prices captured either from the New York Stock Exchange (NYSE), NASDAQ Capital Markets (NASDAQ), and or REFINITIV. Typically, all sources (NYSE, NASDAQ, or REFINITIV) may reflect stock prices with a delay which could be a lag of 15-20 minutes. There can be no assurance that future results or events will be consistent with the information provided in the report. The information is subject to change without any prior notice.
AU




 Please wait processing your request...
Please wait processing your request...