Ironwood Pharmaceuticals Inc
Ironwood Pharmaceuticals, Inc. (NASDAQ: IRWD) is a gastrointestinal (GI) healthcare company engaged in advancing the treatment of GI diseases and redefining the standard of care for GI patients. It is engaged in the development of LINZESS (linaclotide) for adults with irritable bowel syndrome with constipation (IBS-C) or chronic idiopathic constipation (CIC).
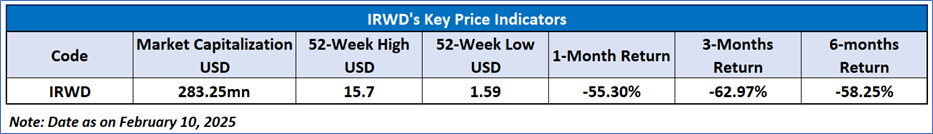
Recent Business and Financial Updates
- Strong Performance of LINZESS and Market Demand: Ironwood Pharmaceuticals, Inc., known for its focus on gastrointestinal treatments, disclosed its financial and operational outcomes for the third quarter of 2024. The flagship product, LINZESS, exhibited robust growth in prescription demand, with both extended units and new-to-brand prescriptions rising by 13% compared to the previous year. Despite facing pricing pressures in 2024, Ironwood has upheld its financial projections for the year. CEO Tom McCourt has confirmed the company's dedication to submitting the New Drug Application (NDA) for apraglutide by the first quarter of 2025, aiming for it to be the premier treatment option for adults with short bowel syndrome (SBS) dependent on parenteral support upon approval.
- Financial Performance and Revenue Analysis: For the third quarter, Ironwood's total revenue was reported at USD91.6 million, a decrease from the USD113.7 million recorded in the same quarter of the previous year, mainly due to reduced income from collaborative arrangements. The company's net income was USD3.6 million, significantly lower than the USD15.3 million from the year before. Adjusted EBITDA was USD26.2 million, down from USD49.1 million, and non-GAAP net income per share was USD0.02, showing a decline from USD0.14 in the prior year's quarter.
- S. LINZESS Market Performance and Collaboration with AbbVie: During the third quarter, LINZESS saw a total prescription demand of 54 million capsules, a 13% increase year-over-year according to IQVIA data. However, U.S. net sales of LINZESS, as reported by collaborator AbbVie Inc., decreased by 19% to USD225.5 million. The commercial margin for LINZESS fell to 65% from 72% in the previous year. The net profit from the U.S. collaboration on the LINZESS brand, post commercial and R&D expenses, was USD139.6 million, down from USD192.0 million. Ironwood recognized USD88.9 million in collaboration revenue, which included a USD5.8 million favorable adjustment concerning LINZESS gross-to-net reserves.
- Pipeline Developments and Strategic Initiatives: Ironwood Pharmaceuticals has progressed with its development pipeline, particularly focusing on apraglutide, a next-generation GLP-2 analog intended for SBS patients needing parenteral support. The company is on schedule with its NDA submission to the FDA and international regulatory filings. Strategically, Ironwood decided against pursuing the exclusive license for CNP-104, thus ending its partnership with COUR Pharmaceuticals. Additionally, the recruitment for the Phase II study of IW-3300, aimed at treating visceral pain, has been paused, with plans to analyze data once current participants conclude their 12-week treatment.
- Financial Position and Credit Facility Expansion: In September 2024, Ironwood amended its revolving credit agreement, boosting the facility limit from USD500 million to USD550 million and extending the due date. By the end of the third quarter, Ironwood had USD88.2 million in cash and cash equivalents. The company settled USD25.0 million of its credit facility, leaving a balance of USD400 million. Operational cash flow for the quarter was USD9.9 million, a reduction from the USD32.5 million reported in the prior year.
- Financial Outlook and Guidance for 2024: Ironwood has maintained its financial outlook for 2024, forecasting U.S. LINZESS net sales to be within USD900 million to USD950 million. Total revenue is projected to be between USD350 million and USD375 million, with an adjusted EBITDA expected to surpass USD75 million. The company's strategy continues to emphasize growth in its core gastrointestinal portfolio alongside advancing key pipeline projects to deliver long-term shareholder value.
Technical Observation (on the daily chart):
The Relative Strength Index (RSI) over a 14-day period stands at a value of 21.69, currently inside the oversold zone, with expectations of a consolidation or a recovery after such a hefty correction. In case of a recovery USD 2.00 levels has to break on the upside for a decent momentum on the upside. Additionally, the stock's current positioning is below both the 50-period SMA and 200-period SMA, which may serve as dynamic short to medium-term resistance levels.
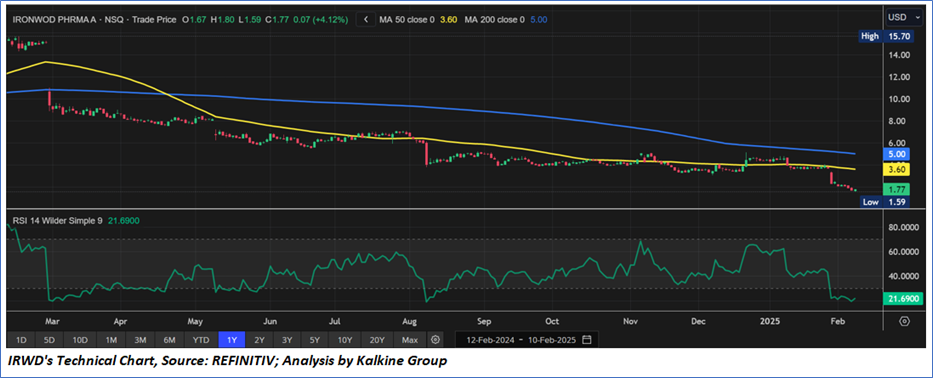
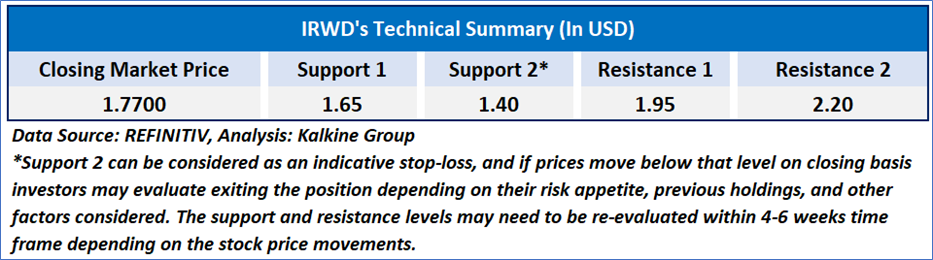
As per the above-mentioned price action, recent key business and financial updates, momentum in the stock over the last month, and technical indicators analysis, a ‘Watch’ rating has been given for Ironwood Pharmaceuticals, Inc. (NASDAQ: IRWD) at the closing price of USD 1.77, as of February 10, 2025.
Individuals can evaluate the stock based on the support and resistance levels provided in the report in case of keen interest taking into consideration the risk-reward scenario.
Markets are trading in a highly volatile zone currently due to certain macro-economic issues and prevailing geopolitical tensions. Therefore, it is prudent to follow a cautious approach while investing.
Related Risk: This report may be looked at from a high-risk perspective and a recommendation is provided for a short duration. This report is solely based on technical parameters, and the fundamental performance of the stocks has not been considered in the decision-making process. Other factors which could impact the stock prices include market risks, regulatory risks, interest rates risks, currency risks, social and political instability risks etc.
Note 1: Past performance is not a reliable indicator of future performance.
Note 2: The reference date for all price data, currency, technical indicators, support, and resistance level is February 10, 2025. The reference data in this report has been partly sourced from REFINITIV.
Note 3: Investment decisions should be made depending on an individual's appetite for upside potential, risks, holding duration, and any previous holdings. An 'Exit' from the stock can be considered if the Target Price mentioned as per the Valuation and or the technical levels provided has been achieved and is subject to the factors discussed above.
Note 4: Target Price refers to a price level that the stock is expected to reach as per the relative valuation method and or technical analysis taking into consideration both short-term and long-term scenarios.
Note 5: ‘Kalkine reports are prepared based on the stock prices captured either from the New York Stock Exchange (NYSE), NASDAQ Capital Markets (NASDAQ), and or REFINITIV. Typically, all sources (NYSE, NASDAQ, or REFINITIV) may reflect stock prices with a delay which could be a lag of 15-20 minutes. There can be no assurance that future results or events will be consistent with the information provided in the report. The information is subject to change without any prior notice.
AU




 Please wait processing your request...
Please wait processing your request...