Ironwood Pharmaceuticals Inc
Ironwood Pharmaceuticals, Inc. (NASDAQ: IRWD) is a gastrointestinal (GI) healthcare company engaged in advancing the treatment of GI diseases and redefining the standard of care for GI patients. It is engaged in the development of LINZESS (linaclotide) for adults with irritable bowel syndrome with constipation (IBS-C) or chronic idiopathic constipation (CIC).
Recent Business and Financial Updates
- Financial Performance Overview for Q1 2025: Ironwood Pharmaceuticals, Inc. reported a Q1 2025 total revenue of USD 41.1 million, a decline from USD 74.9 million in Q1 2024, primarily due to a change in AbbVie’s gross-to-net rebate reserves affecting LINZESS U.S. net sales, as announced on May 7, 2025. The company recorded a GAAP net loss of USD 37.4 million (USD 0.23 per share), compared to a USD 4.2 million loss (USD 0.03 per share) in Q1 2024, with adjusted EBITDA dropping to a loss of USD 4.7 million from a USD 21.1 million gain, reflecting higher costs and expenses of USD 70.3 million, up from USD 63.9 million. Despite these challenges, Ironwood raised its 2025 adjusted EBITDA guidance to over USD 105 million, signaling confidence in its full-year financial outlook.
- LINZESS Performance and U.S. Brand Collaboration: LINZESS, Ironwood’s flagship product, demonstrated robust prescription demand growth of 8% year-over-year in Q1 2025, with 53 million capsules prescribed, aligning with full-year expectations, though U.S. net sales fell 46% to USD 138.5 million from USD 256.6 million in Q1 2024 due to rebate adjustments. The commercial margin for LINZESS decreased to 52% from 71%, and net profit from the U.S. brand collaboration with AbbVie dropped 62% to USD 65.9 million, reflecting the impact of the rebate reserve change, which Ironwood expects to offset in subsequent quarters with no full-year impact. This sustained demand underscores LINZESS’s market strength, a key factor for assessing Ironwood’s revenue stability.
- Progress on Apraglutide Development: Ironwood announced in April 2025 that, following FDA discussions, a confirmatory Phase 3 trial will be required for apraglutide’s approval in short bowel syndrome (SBS) with intestinal failure (IF), prompting the company to continue its long-term extension trial and collaborate with the FDA on trial design and regulatory pathways. The STARS Phase 3 trial, the largest SBS-IF study to date, and its extension trial showed promising results, with 27 apraglutide-dosed patients achieving enteral autonomy, reinforcing its potential as a transformative therapy. These developments highlight Ironwood’s commitment to advancing its pipeline, a critical consideration for long-term growth prospects.
- Strategic Initiatives to Enhance Stockholder Value: Ironwood engaged Goldman Sachs & Co. LLC in April 2025 to explore strategic alternatives aimed at maximizing stockholder value, signaling a proactive approach to optimizing its business structure and market position. This initiative, combined with the company’s focus on advancing apraglutide and maintaining LINZESS’s growth trajectory, reflects a strategic effort to enhance shareholder returns amidst operational challenges. Investors should note this as a potential catalyst for future value creation, depending on the outcomes of the strategic review.
- Cash Flow and Financial Position: Ironwood ended Q1 2025 with USD 108.5 million in cash and cash equivalents, up from USD 88.6 million at the end of 2024, generating USD 20.0 million in cash from operations, though down from USD 45.0 million in Q1 2024, due to increased expenses and revenue adjustments. The company’s financial position, supported by its LINZESS collaboration revenue of USD 38.8 million, provides a foundation for funding its pipeline development and strategic initiatives. This cash position and operational cash flow are essential for evaluating Ironwood’s ability to sustain its growth strategy as of June 5, 2025.
- 2025 Financial Guidance and Outlook: Ironwood reaffirmed its 2025 guidance, expecting U.S. LINZESS net sales of USD 800–850 million, total revenue of USD 260–290 million, and high single-digit prescription demand growth for LINZESS, though tempered by expected price erosion due to Medicare Part D redesign. The raised adjusted EBITDA guidance of over USD 105 million reflects optimism in achieving profitability targets despite Q1 challenges, providing investors with a clearer view of the company’s financial trajectory. As of June 5, 2025, Ironwood’s focus on LINZESS demand growth, pipeline advancement, and strategic alternatives positions it to navigate near-term challenges while pursuing long-term value creation.
Technical Observation (on the daily chart):
The 14-day Relative Strength Index (RSI) is currently at 38.62, currently recovering from oversold levels, with the expectations of consolidation or some upward momentum once important resistance of USD 0.65-USD0.70 is broken on the upside. In addition, the current price is below both the 50-day Simple Moving Averages (SMAs) and 200-day SMA, which may work as medium to long term resistance levels.
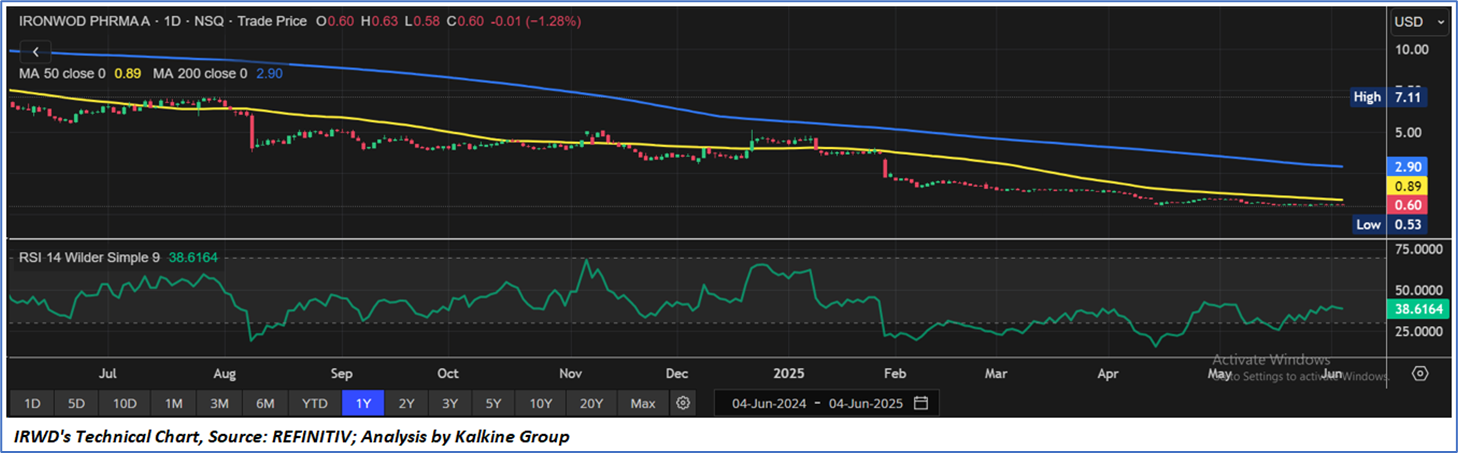
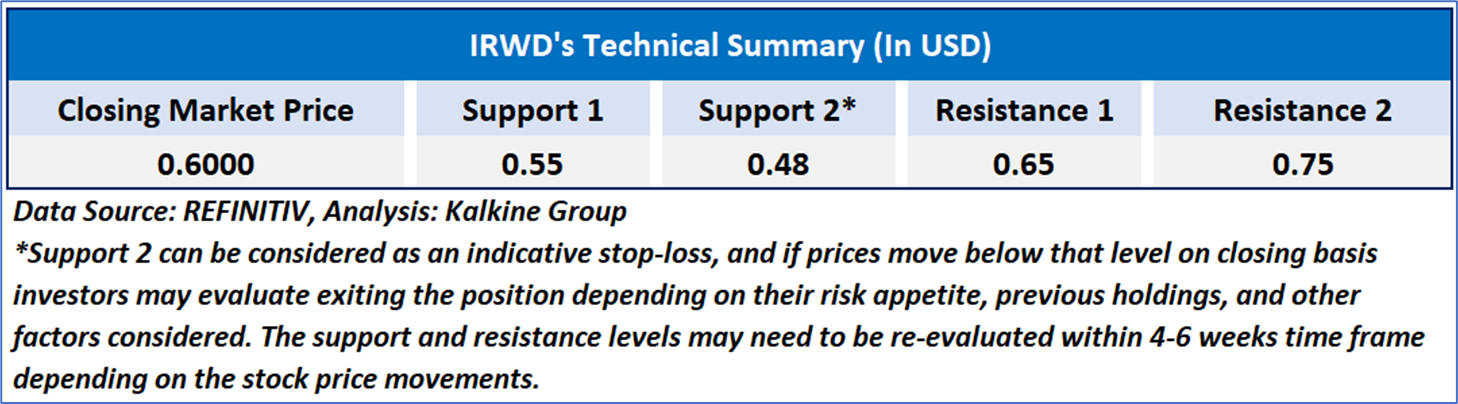
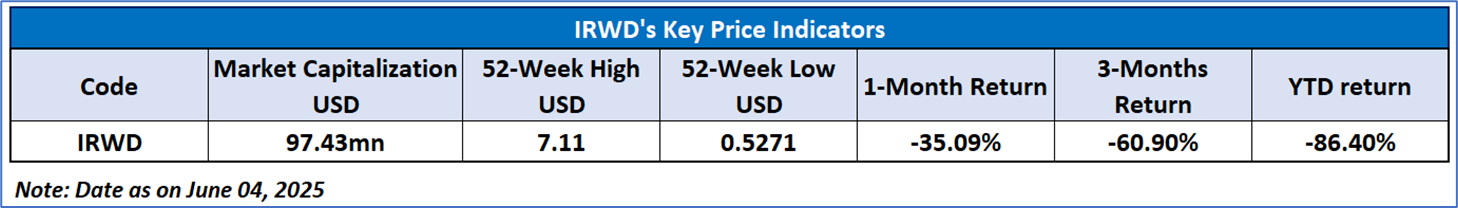
As per the above-mentioned price action, recent key business and financial updates, momentum in the stock over the last month, and technical indicators analysis, a ‘WATCH’ rating has been given Ironwood Pharmaceuticals, Inc. (NASDAQ: IRWD) at the closing price of USD0.60, as of June 04, 2025.
Individuals can evaluate the stock based on the support and resistance levels provided in the report in case of keen interest taking into consideration the risk-reward scenario.
Markets are trading in a highly volatile zone currently due to certain macro-economic issues and prevailing geopolitical tensions. Therefore, it is prudent to follow a cautious approach while investing.
Related Risk: This report may be looked at from a high-risk perspective and a recommendation is provided for a short duration. This report is solely based on technical parameters, and the fundamental performance of the stocks has not been considered in the decision-making process. Other factors which could impact the stock prices include market risks, regulatory risks, interest rates risks, currency risks, social and political instability risks etc.
Note 1: Past performance is not a reliable indicator of future performance.
Note 2: The reference date for all price data, currency, technical indicators, support, and resistance level is June 04, 2025. The reference data in this report has been partly sourced from REFINITIV.
Note 3: Investment decisions should be made depending on an individual's appetite for upside potential, risks, holding duration, and any previous holdings. An 'Exit' from the stock can be considered if the Target Price mentioned as per the Valuation and or the technical levels provided has been achieved and is subject to the factors discussed above.
Note 4: Target Price refers to a price level that the stock is expected to reach as per the relative valuation method and or technical analysis taking into consideration both short-term and long-term scenarios.
Note 5: ‘Kalkine reports are prepared based on the stock prices captured either from the New York Stock Exchange (NYSE), NASDAQ Capital Markets (NASDAQ), and or REFINITIV. Typically, all sources (NYSE, NASDAQ, or REFINITIV) may reflect stock prices with a delay which could be a lag of 15-20 minutes. There can be no assurance that future results or events will be consistent with the information provided in the report. The information is subject to change without any prior notice.
AU




 Please wait processing your request...
Please wait processing your request...