Mind Medicine (MindMed) Inc
Mind Medicine (MindMed) Inc. (NASDAQ: MNMD) is a clinical-stage biopharmaceutical company, which is engaged in developing products to treat brain health disorders. It is developing a pipeline of product candidates, with and without acute perceptual effects, targeting neurotransmitter pathways. This specifically includes pharmaceutically optimized product candidates derived from the psychedelic and empathogen drug classes, including MM-120 and MM-402, the Company's product candidates.
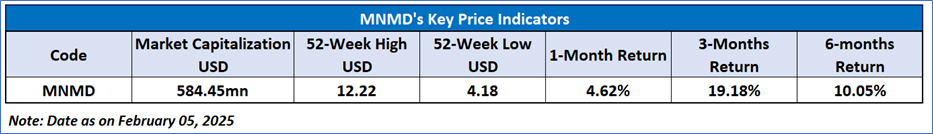
Recent Business and Financial Updates
- Overview and Key Milestones: MindMed Reports its Third Quarter 2024 financial results and provides significant business updates as it continues to advance its clinical-stage biopharmaceutical programs for brain health disorders. Notable highlights include the anticipated initiation of the Phase 3 Voyage study of MM120 ODT in Generalized Anxiety Disorder (GAD) in Q4 2024, with 12-week topline data expected in the first half of 2026, and plans to launch additional Phase 3 studies—the Panorama study in GAD and the Emerge study in Major Depressive Disorder (MDD)—in the coming months. The company’s robust financial position, with cash and cash equivalents of USD 295.3 million as of September 30, 2024, is projected to fund operations through 2027 and extend well beyond the first Phase 3 data readout.
- MM120 Program for Generalized Anxiety Disorder: MindMed is progressing with its MM120 ODT program for the treatment of GAD through two pivotal Phase 3 studies. The Voyage study (MM120-300) is on track to initiate in the fourth quarter of 2024 and is expected to enroll approximately 200 U.S. participants, randomized to receive either 100 µg of MM120 ODT or placebo. Complementing this is the Panorama study (MM120-301), anticipated to enroll around 240 participants with a randomization scheme of 5:2:5 for the MM120 ODT 100 µg, MM120 ODT 50 µg, or placebo groups. Both studies will assess the primary endpoint of change from baseline in Hamilton Anxiety Rating Scale (HAM-A) scores at Week 12 and incorporate an adaptive design with interim sample size re-estimation to maintain statistical power.
- MM120 Program for Major Depressive Disorder: In addition to its GAD program, MindMed is advancing the MM120 ODT program for Major Depressive Disorder (MDD) through the Emerge study (MM120-310). This Phase 3 trial, designed similarly to the GAD studies, will consist of a 12-week, randomized, double-blind, placebo-controlled period (Part A), followed by a 40-week extension (Part B) during which participants may receive open-label treatment with MM120 ODT. The study is expected to enroll at least 140 participants, with the primary endpoint measured as the change from baseline in Montgomery-Åsberg Depression Rating Scale (MADRS) scores at Week 6. Initiation of the Emerge study is anticipated in the first half of 2025, with topline data from the 12-week period expected in the second half of 2026.
- MM402 Development for Autism Spectrum Disorder: MindMed also reported progress on its MM402 program, an initiative focused on addressing Autism Spectrum Disorder (ASD). In October 2024, the company successfully completed a Phase 1 single-ascending dose study in healthy adult volunteers, which evaluated the tolerability, pharmacokinetics, and pharmacodynamics of MM402 (R(-)-MDMA). These early-stage results pave the way for further studies to explore the potential of MM402 in the treatment of ASD, with the timing and scope of future research to be determined as the program evolves.
- Financial Position and Cash Flow: For the third quarter of 2024, MindMed reported a cash and cash equivalents balance of USD 295.3 million, a substantial increase from USD 99.7 million as of December 31, 2023. This robust liquidity is expected to fund the company’s operations into 2027 and provide a significant runway beyond the anticipated Phase 3 topline data readout for MM120 ODT in GAD. During the nine-month period ended September 30, 2024, net cash used in operating activities was USD 53.8 million, compared to USD 43.8 million during the corresponding period in 2023, reflecting increased investments in advancing clinical development.
- Operating Expenses and Net Loss: MindMed’s research and development expenses for Q3 2024 amounted to USD 17.2 million, marking an increase of USD 4.0 million over the prior year’s quarter. This rise was driven by higher expenditures related to the MM120 program, additional costs associated with the MM402 program, increased internal personnel costs, and expanded preclinical activities. In contrast, general and administrative expenses decreased to USD 7.6 million from USD 8.4 million, primarily due to lower legal and commercial spending, partially offset by an increase in stock-based compensation. Overall, the net loss for Q3 2024 was USD 13.7 million, a reduction of USD 4.2 million from USD 17.9 million in Q3 2023, reflecting the company’s efforts to manage costs while progressing its clinical programs.
Technical Observation (on the daily chart):
The Relative Strength Index (RSI) over a 14-day period stands at a value of 58.92, currently upward moving, with expectations of a consolidation or an upward momentum once USD 8.00-USD9.00 resistance is broken on the upside. Additionally, the stock's current positioning is above both the 50-period SMA and 200-period SMA, which may serve as dynamic short to medium-term support levels.
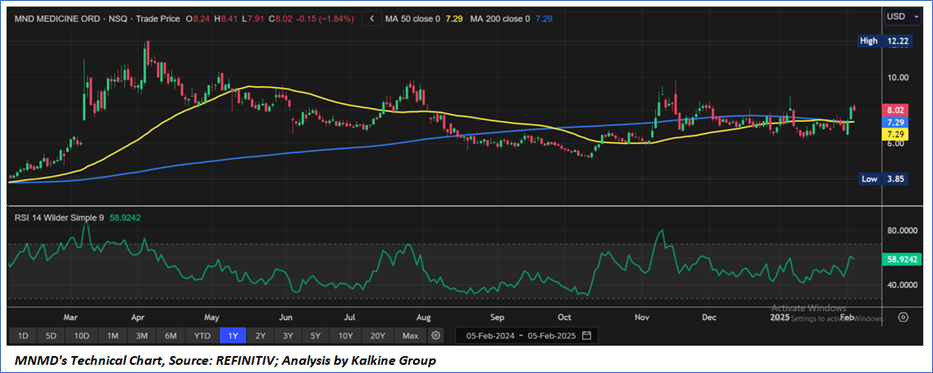
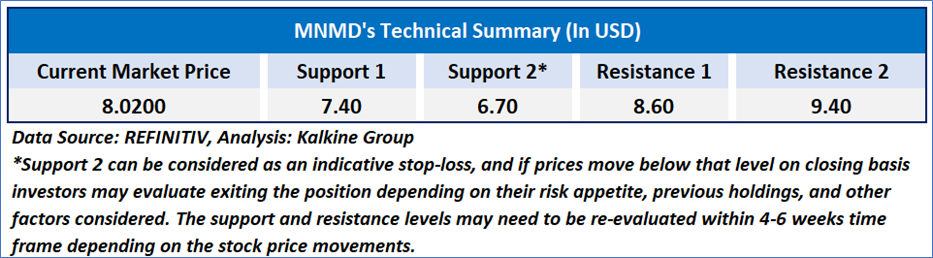
As per the above-mentioned price action, recent key business and financial updates, momentum in the stock over the last month, and technical indicators analysis, a ‘WATCH`’ rating has been given for Mind Medicine (MindMed) Inc. (NASDAQ: MNMD) at the current price of USD 8.02 as of February 05, 2025, at 07:10 am PST.
Individuals can evaluate the stock based on the support and resistance levels provided in the report in case of keen interest taking into consideration the risk-reward scenario.
Markets are trading in a highly volatile zone currently due to certain macro-economic issues and prevailing geopolitical tensions. Therefore, it is prudent to follow a cautious approach while investing.
Related Risk: This report may be looked at from a high-risk perspective and a recommendation is provided for a short duration. This report is solely based on technical parameters, and the fundamental performance of the stocks has not been considered in the decision-making process. Other factors which could impact the stock prices include market risks, regulatory risks, interest rates risks, currency risks, social and political instability risks etc.
Note 1: Past performance is not a reliable indicator of future performance.
Note 2: The reference date for all price data, currency, technical indicators, support, and resistance level is February 05, 2025. The reference data in this report has been partly sourced from REFINITIV.
Note 3: Investment decisions should be made depending on an individual's appetite for upside potential, risks, holding duration, and any previous holdings. An 'Exit' from the stock can be considered if the Target Price mentioned as per the Valuation and or the technical levels provided has been achieved and is subject to the factors discussed above.
Note 4: Target Price refers to a price level that the stock is expected to reach as per the relative valuation method and or technical analysis taking into consideration both short-term and long-term scenarios.
Note 5: ‘Kalkine reports are prepared based on the stock prices captured either from the New York Stock Exchange (NYSE), NASDAQ Capital Markets (NASDAQ), and or REFINITIV. Typically, all sources (NYSE, NASDAQ, or REFINITIV) may reflect stock prices with a delay which could be a lag of 15-20 minutes. There can be no assurance that future results or events will be consistent with the information provided in the report. The information is subject to change without any prior notice.
AU




 Please wait processing your request...
Please wait processing your request...