Alto Neuroscience, Inc
Alto Neuroscience, Inc (NYSE: ANRO) is a clinical-stage biopharmaceutical company dedicated to transforming psychiatry by using insights into individual neurobiology to create personalized and more effective treatments.
Positive Growth Prospects
- Robust and Diversified Clinical Pipeline: Alto Neuroscience is making significant strides in advancing its clinical-stage pipeline, which targets high unmet needs in neuropsychiatric disorders through a precision medicine approach. The company is actively conducting four Phase 2 clinical trials: ALTO-100 for bipolar depression (BPD), ALTO-300 for adjunctive major depressive disorder (MDD), ALTO-203 for MDD with anhedonia, and ALTO-101 for cognitive impairment associated with schizophrenia (CIAS). The company's strategic use of EEG-based and cognitive biomarkers to identify responsive subpopulations exemplifies its commitment to personalized treatment strategies. This approach not only enhances the potential for clinical success but also distinguishes Alto in the competitive neuropsychiatric drug development space.
- Strategic Use of Biomarkers and Technological Innovation: A notable strength of Alto lies in its biomarker-driven development strategy. For instance, its use of EEG-based biomarkers to identify placebo responders and dopamine-related biomarkers to define treatment-sensitive subgroups represents a cutting-edge application of neurobiological insights in trial design. Such techniques have demonstrated promising results, such as improving treatment signal detection and increasing statistical power in trials. The mechanistic insights linking drug candidates like ALTO-300 to specific biomarker profiles further support the rationale for patient stratification, potentially leading to faster approvals and better therapeutic outcomes.
- Financial Stability and Forward-Looking Outlook: Alto ended Q1 2025 with USD 161.3 million in cash, cash equivalents, and restricted cash, a decrease of just USD 7.4 million from the previous quarter. The company expects this cash reserve to support its operations into 2028, providing a solid financial runway for executing its ongoing trials and advancing its pipeline. Importantly, Alto is efficiently managing its R&D budget, with consistent expenditures year-over-year, while strategically increasing G&A spending to support its transition into a more complex, publicly traded organization. These financial indicators suggest prudent fiscal management amid an ambitious development agenda.
- Active Engagement in Scientific and Investor Communities: Alto is maintaining strong visibility within both the scientific and investor communities. The company has actively presented its research at major psychiatric and neuroscience conferences, such as SOBP and SIRS, and is scheduled to present at several key investor conferences, including ASCP and BIO. This demonstrates Alto’s commitment to transparency, thought leadership, and investor engagement. Such visibility is essential for fostering partnerships, attracting new investors, and affirming the company's leadership in precision psychiatry.
Growth Challenges
- Prolonged Clinical Timelines and Delayed Data Readouts: While Alto boasts a promising pipeline, the timeline for meaningful clinical readouts is relatively extended. For instance, topline data for ALTO-100 in BPD and ALTO-300 in MDD are not expected until the second half of 2026 and mid-2026, respectively. These extended timelines increase the risk of clinical or operational setbacks and may test investor patience in a market that often favors nearer-term catalysts. The longer development horizon could also delay revenue generation and strategic milestones like potential partnerships or regulatory submissions.
- Modest Progress in Biomarker Validation and Early-Stage Data: Although Alto is at the forefront of biomarker-based neuropsychiatric drug development, much of its data remains early-stage or exploratory in nature. For example, the EEG-based placebo response biomarker, while promising, is still being prospectively validated in ongoing trials. Similarly, ALTO-203’s proof-of-concept trial is not powered to demonstrate statistical significance on traditional clinical scales like MADRS. This reliance on exploratory endpoints and novel biomarkers, while innovative, introduces uncertainty around clinical translation and regulatory acceptance.
- Increased Operating Costs and Rising Net Loss: Alto reported a net loss of USD 15.2 million in Q1 2025, compared to USD 13.4 million in Q1 2024, reflecting increased spending, particularly in general and administrative expenses. While the increase is largely attributed to expanded clinical development and the operational demands of being a public company, it signals a growing cost base that could pressure financial sustainability over time. If clinical timelines slip or trial results disappoint, the company's increasing burn rate could necessitate future capital raises, potentially diluting existing shareholders.
- Regulatory and Competitive Challenges in the U.S. Market: Despite agomelatine (ALTO-300) being approved in Europe and Australia, it remains unapproved in the United States, which presents regulatory risk. Although Alto is developing the lower 25mg dose, which appears to mitigate liver enzyme issues, the path to U.S. approval remains uncertain. Furthermore, the neuropsychiatric space is highly competitive, with multiple companies pursuing treatments for MDD and schizophrenia. Alto's reliance on biomarker-defined subgroups may narrow its target patient population, potentially limiting commercial scale if broader efficacy is not demonstrated.
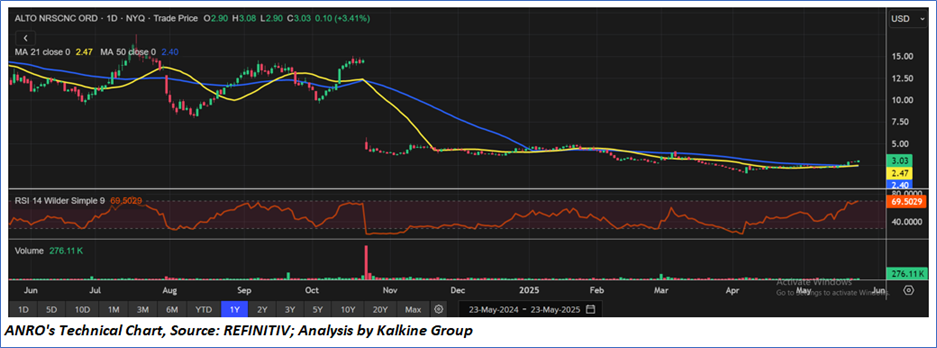
Technical Observation (on the daily chart):
Alto Ingredients Inc. is showing early signs of a potential trend reversal after a prolonged downtrend and multi-month consolidation. The stock recently broke above key moving averages, with a bullish crossover of the 21-day over the 50-day MA, and the RSI is nearing overbought territory at 69.5—indicating strong momentum. However, volume remains relatively low.
Alto Neuroscience presents a compelling yet cautiously optimistic investment profile, marked by a strong cash position, innovative use of biomarkers, and a diversified pipeline targeting significant unmet needs in neuropsychiatric disorders. However, the company faces challenges such as extended clinical timelines, increased operational costs, and the inherent risks of early-stage biomarker validation. While its precision psychiatry approach and scientific engagement are commendable, clinical and regulatory uncertainties—especially in the U.S. market—temper the near-term outlook. Overall, Alto offers high potential upside, but with notable execution and timing risks.
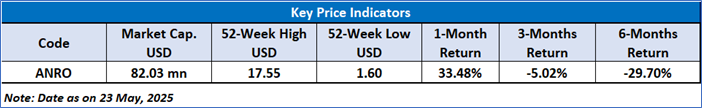
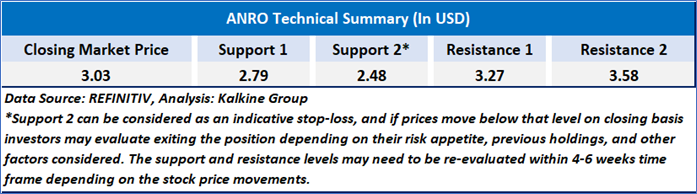
As per the above-mentioned price action, recent key business and financial updates, momentum in the stock over the last month, and technical indicators analysis, a ‘Watch’ rating has been given to Alto Neuroscience, Inc (NYSE: ANRO) at the closing market price of USD 3.03 as of May 23,2025.
Individuals can evaluate the stock based on the support and resistance levels provided in the report in case of keen interest taking into consideration the risk-reward scenario.
Markets are trading in a highly volatile zone currently due to certain macro-economic issues and prevailing geopolitical tensions. Therefore, it is prudent to follow a cautious approach while investing.
Related Risk: This report may be looked at from a high-risk perspective and a recommendation is provided for a short duration. This report is solely based on technical parameters, and the fundamental performance of the stocks has not been considered in the decision-making process. Other factors which could impact the stock prices include market risks, regulatory risks, interest rates risks, currency risks, social and political instability risks etc.
Note 1: Past performance is not a reliable indicator of future performance.
Note 2: The reference date for all price data, currency, technical indicators, support, and resistance level is May 23,2025. The reference data in this report has been partly sourced from REFINITIV.
Note 3: Investment decisions should be made depending on an individual's appetite for upside potential, risks, holding duration, and any previous holdings. An 'Exit' from the stock can be considered if the Target Price mentioned as per the Valuation and or the technical levels provided has been achieved and is subject to the factors discussed above.
Note 4: Target Price refers to a price level that the stock is expected to reach as per the relative valuation method and or technical analysis taking into consideration both short-term and long-term scenarios.
Note 5: ‘Kalkine reports are prepared based on the stock prices captured either from the New York Stock Exchange (NYSE), NASDAQ Capital Markets (NASDAQ), and or REFINITIV. Typically, all sources (NYSE, NASDAQ, or REFINITIV) may reflect stock prices with a delay which could be a lag of 15-20 minutes. There can be no assurance that future results or events will be consistent with the information provided in the report. The information is subject to change without any prior notice.
AU




 Please wait processing your request...
Please wait processing your request...